Such a system provides a source for assays in human hematopoietic and immune system related functions in vivo, identification of novel therapies and vaccines. However, no in vivo system exists to determine the molecular aspects of human immune cell-to-cell interactions and its regulation. Such communication is critical for the survival of such organisms, as these same proteins are suggested to be an important determinant of transplant engraftment. Further, these same immune cells have been found to possess a self-awareness property by virtue of, inter alia, regulatory membrane proteins that regulate cell-to-cell interactions. Getting Started with MacVector: An overview of primer design workflows in MacVector.The immune system is composed of several different cell types that are involved in multiple highly regulated processes and together generate immune responses that are effective in eliminating foreign proteins.Melissa Caimano on HOW DO I video guides to common molecular biology workflows.admin on HOW DO I video guides to common molecular biology workflows.mariam abdelmalak on Major release details – Summary.Brian on Designing primers and documenting In-Fusion Cloning with MacVector.Chris on Designing primers and documenting In-Fusion Cloning with MacVector.
MacVectorTip: displaying CRISPR PAM Sites on a sequence.MacVectorTip: Sign up for an NCBI API key to speed up BLAST results.MacVectorTip: Designing Primers for Gibson Assembly.MacVectorTip: Simulating mixed plasmid populations in agarose gels.
#Macvector inc how to
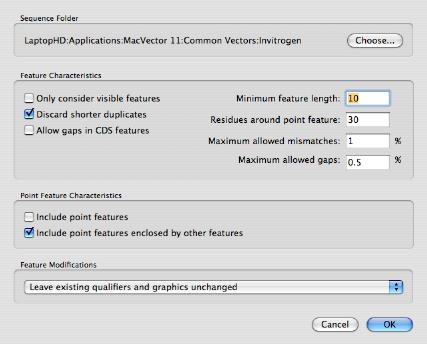
MacVectorTip: How to find Restriction Enzymes that only cut outside of a specific region.Track 5 shows the banding pattern for a 50:50 mix of the two plasmids (not the non equip-molar intensity of the variant bands) and track 6 shows the difference in band intensity when the mutant plasmid represents just one third of the molecules. In gel tracks 3 and 4 you can see the different banding patterns between pBR322 and pBR322+. (Note that by default MacVector only shows a maximum of 8 cuts per enzyme so we increased this to unlimited in the RE Picker to ensure the FspI sites were displayed). Finally we used File | New Agarose Gel to create an empty gel, then selected FspI sites from each of the four molecules and dragged them onto separate lanes in the agarose gel. Then we added a second copy of pBR322 into one of the EcoRI sites to create a molecule with two copies of pBR322 and one copy of pBR322+ (Hybrid–2+1). We saved this molecule with the two directly repeated plasmids “ Hybrid–1+1”. We then joined a copy of both plasmids by selecting the unique EcoRI site in pBR322, selecting Edit | Copy, then selecting the EcoRI site in pBR322+ and choosing Edit | Paste. We used pBR322 and introduced a fifth FspI site by changing the T at 2788 to a C, creating pBR322+.
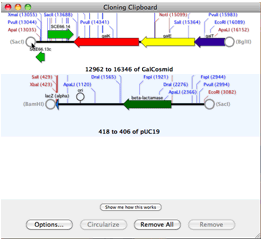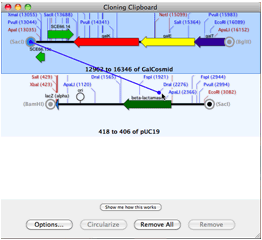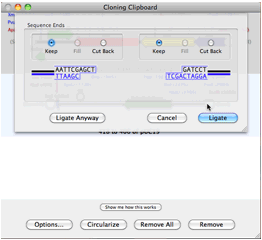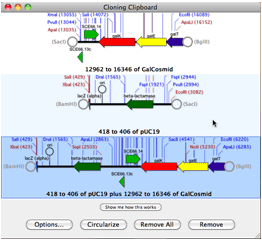
You can simulate this in MacVector by creating concatenated fake plasmids counting the two variant plasmids. You might come across this type of scenario if you have been making site-specific mutations and introducing new restriction sites into a vector where only some of the resulting plasmids might have acquired the extra site. We had a recent support call this week from somebody who believed from their agarose gels that they had a mixed population of plasmids from an experiment and wanted to document and determine the banding pattern using MacVector’s agarose gel simulation.
